Body-Centered Tetragonal (abbreviated BCT in this article) is one of the advanced crystal structures, so if you’re searching for information about it, I’m assuming you’re a somewhat advanced student in materials science. I’ll give you the facts quickly and with little explanation. If you are new to materials science and don’t know what something means, you can check out my articles on the FCC, BCC, or HCP crystals, which explain terminology more slowly.
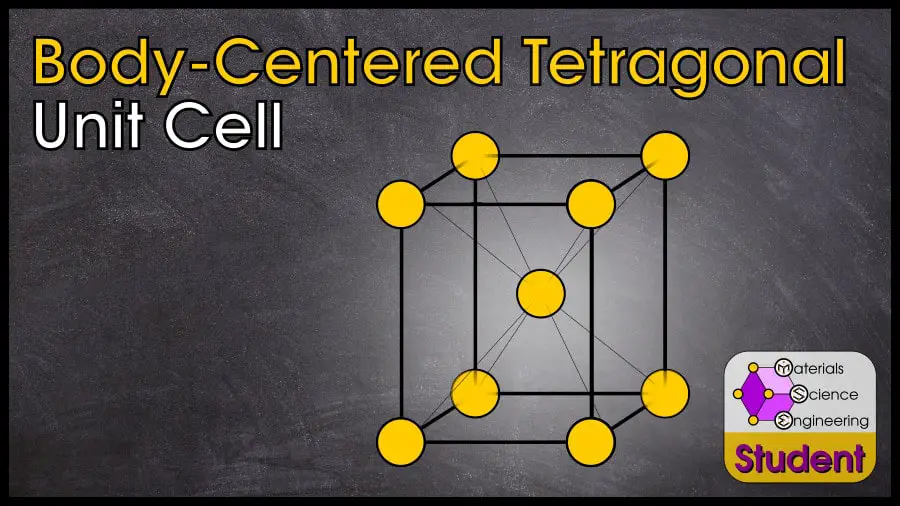
The body-centered tetragonal crystal structure is based on the Bravais lattice of the same name, with 1 atom per lattice point. 8 corner atoms make up a tetragon (which is a cube with one side of a different length), and 1 atom is in the volumetric center. (It may also be interesting to note that body-centered tetragonal and face-centered tetragonal are identical, so we just use BCT). The body-centered tetragonal crystal structure belongs to space group #139 or I4/mmm, Strukturbericht A6 or Aa, and Pearson symbol tI2. Depending on the c/a ratio, there are two prototypes for BCT: alpha-protactinium is the prototype when the c/a ratio is close to one, and indium is the prototype when the c/a ratio is close to .
The body-centered tetragonal unit cell can be imagined as a cube that is slightly taller or shorter in one direction, with an atom on each corner and in the very center. Body-Centered Tetragonal has 2 atoms per unit cell and Coordination Number CN = 8 to 12 (with a “perfect” CN= 10).
Outline
Common Examples of Body-Centered Tetragonal Materials
Indium, protactinium, and beta-mercury exhibit the body-centered tetragonal structure near standard pressure and temperature (STP). Mercury is of course a liquid, but the first solid it freezes into is BCT.
The most common body-centered tetragonal crystal you may have heard of is martensite in steel. However, classifying steel martensite is a bit tricky since it exists because of carbon in interstitial sites. If you looked at only the iron atoms, it would be BCT, but you can’t ignore the carbon. Martensite has BCT symmetry and a BCT Bravais lattice, but it is NOT the exact crystal structure described in this article.
Body-Centered Tetragonal Coordination Number
In the cubic BCC, there are 8 nearest-neighbors, for a coordination number (CN) of 8. In FCC, there are 12 nearest-neighbors.
When distorting one direction of a cube, the body-centered tetragonal bridges the space between BCC and FCC. Therefore, it’s possible for BCT to have a coordination number between 8 and 12, and it’s theoretically possible to have a coordination number lower than either BCT or FCC.
Measuring coordination number gets tricky in noncubic systems.
If the c/a ratio is exactly , the structure has an exact CN = 10. This c/a ratio is 8.16, which is similar to the experimental c/a ratio of protactinium.
Body-Centered Tetragonal Lattice Constants
Since a tetragon has three vectors at 90º to each other, where 2 are the same length and one is different, there are two lattice constants: a, b (which is also a), and c.
By convention, crystallographers use a and the c/a ratio to model most crystals that have two parameters: a and the c/a ratio.
It’s not really possible to mathematically derive the ideal lattice parameters for BCT because the ideal would actually be BCC or FCC. If you were working with a BCT crystal, you would have to measure the parameters a and c directly.
If you wanted to describe the body-centered tetragonal crystal with math, you would describe the cell with the vectors:
And of course the basis is 0, because there is an exact 1-1 correspondence between the lattice points and atoms.
Body-Centered Tetragonal Atomic Packing Factor
Just as it’s impossible to derive a theoretically ideal lattice parameter for Simple Tetragonal, it’s also impossible to derive a theoretically ideal APF for BCT.
However, you can calculate it if you know your lattice parameters. I’ll go through the calculation with you quickly, although you can follow my detailed article on APF if you’re not sure about something.
To calculate the atomic packing factor (APF), you need to find the portion of space taken up by atoms (as opposed to empty space). We do this calculation by assuming the atoms are hard spheres.
Each unit cell is a rectangular prism with two sides equal (a) and one side different (c). Therefore, the volume of the unit cell is .
If you count the portions of the atoms inside the unit cell, there are 8 atoms on each corner which contribute ⅛ of their volume, and 1 atom on the inside which contributes its full volume. Thus, there are 2 atoms per unit cell.
The volume of a sphere is . We previously established that the volume of the whole cell is
, and so if we know the lattice parameters a and c, and atomic radius r, we can calculate the atomic packing factor.
Primitive Body-Centered Tetragonal Unit Cell
Advanced topic, click to expand!
The conventional BCC cell that I have shown you is a conventional unit cell, not a primitive unit cell. This conventional cell has advantages because it is highly symmetric and easy for humans to understand.
However, when dealing with mathematical descriptions of crystals, it may be easier to describe the unit cell in the smallest form possible. The smallest possible unit cell is called the primitive cell. If you are interested in primitive cells, you can read all about them in this article.
The BCT primitive cell looks like this:
Here are the primitive vectors for the BCT unit cell.
Final Thoughts
The body-centered tetragonal crystal structure closely resembles the body-centered cubic crystal structure, but with 1 axis distorted to be longer or shorter than the other two.
If you know about martensite in steel, you may have heard that this phase is BCT. Remember, BCT can mean a crystal structure or a Bravais lattice. Martensite has a BCT Bravais lattice, but the crystal structure is more complex than the 1-atom-per-lattice point definition of the BCT crystal.
References and Further Reading
If you want to know more about the basics of crystallography, check out this article about crystals and grains.
If you weren’t sure about the difference between crystal structure and Bravais lattice, check out this article.
I also mentioned atomic packing factor (APF) earlier in this article. This is an important concept in your introductory materials science class, so if you want a full explanation of APF, check out this page.
If you’re interested in advanced crystallography or crystallography databases, you may want to check out the AFLOW crystallographic library.
For a great reference for all crystal structures, check out “The AFLOW Library of Crystallographic Prototypes.”
The 14 Basic Crystal Structures
If you want to learn about specific crystal structures, here is a list of my articles about Bravais lattices and some related crystal structures for pure elements. Body-Centered Tetragonal is one of these 14 Bravais lattices and also occurs as a crystal structure.
1. Simple Cubic
2. Face-Centered Cubic
2a. Diamond Cubic
3. Body-Centered Cubic
4. Simple Hexagonal
4a. Hexagonal Close-Packed
4b. Double Hexagonal Close-Packed (La-type)
5. Rhombohedral
5a. Rhombohedral Close-Packed (Sm-type)
6. Simple Tetragonal
7. Body-Centered Tetragonal
7a. Diamond Tetragonal (White Tin)
8. Simple Orthorhombic
9. Base-Centered Orthorhombic
10. Face-Centered Orthorhombic
11. Body-Centered Orthorhombic
12. Simple Monoclinic
13. Base-Centered Monoclinic
14. Triclinic
Other articles in my crystallography series include:
Introduction to Bravais Lattices
What is the Difference Between “Crystal Structure” and “Bravais Lattice”
Atomic Packing Factor
How to Read Miller Indices
How to Read Hexagonal Miller-Bravais Indices
Close-Packed Crystals and Stacking Order
Interstitial Sites
Primitive Cells
How to Read Crystallography Notation
What are Point Groups
List of Point Groups
What are Space Groups
List of Space Groups
The 7 Crystal Systems