
As someone who studies metallurgy for a living, I am often annoyed by the misinformation that is spread about alloys. I can (mostly) forgive movies like Pacific Rim when they suggest that pure metals are somehow stronger than alloys, but I’ve even seen misinformation spread about alloys on “informative” websites. It’s time to set the record straight.
Alloys are metals made of more than one element. That’s it. As long as the final product behaves like a metal, but it’s not elementally pure, it’s an alloy. There are no special rules about solid solution, number of phases, or the ratio of metallic and non-metallic elements. You can also think of alloys as intentionally designed metals.
Alloys, like all metals, are reflective, strong, good conductors, and (usually) ductile. These properties come from metallic bonding.
Outline
Why are Alloys Useful?
By alloying–adding new elements to a pure metal–you can achieve new properties. For example, suppose you mixed lead and tin. Doing this will result in a lower melting point than either pure metal!
Just as alloying decreases the melting point, it increases the material’s strength. Brass and bronze are much stronger than pure copper. Steel (iron alloyed with carbon) is much stronger than pure iron.
Notice that a copper and nickel alloy has higher strength than a simple composite of pure nickel and pure copper.
There are very few situations where a pure metal performs better than an alloy. Usually, lower melting point is good because it requires less energy to process the final product. Higher strength is usually good, because strength is one of a material’s most important properties.
How are Alloys Designed?
Alloys are made by mixing a metal with other elements (usually more metals). This “mixing” can be done by melting the elements together (casting, induction melting, or vacuum arc melting).
A more expensive way of making alloys is called powder metallurgy, which squishes metal particles together and fuses them in a process called “sintering.” Powder metallurgy has many advantages over regular melting because you can combine elements with dissimilar melting points and make sure the mixture is homogeneous. However, it’s not easy to make metal powder!
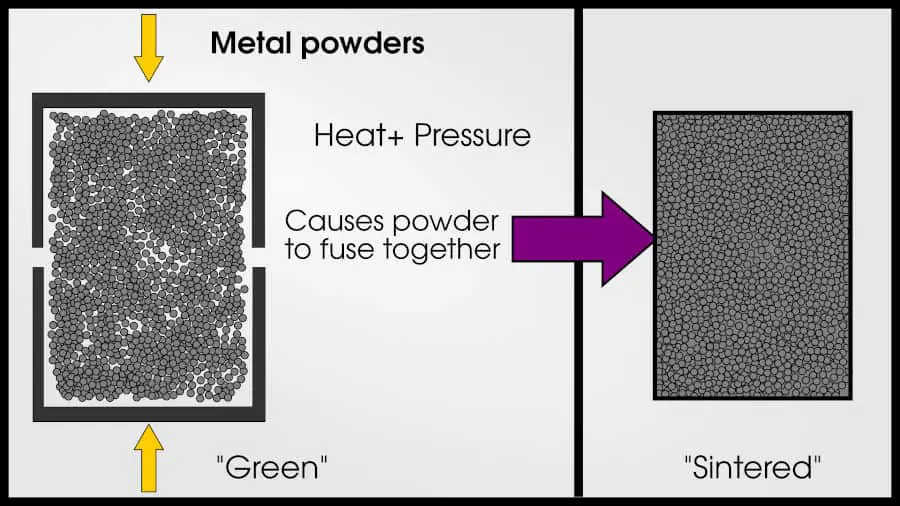
Alloys are typically designed to be in solid solution. It’s possible to have an alloy that is not in solid-solution, but these cases are rare. Generally, if two metals are not in solid solution,they will not have “metallic” properties (and won’t be called “alloys”). Alloys can also have precipitates which are not in solid-solution.
Solid solution means that one phase is dominant. Atoms of the other element are dissolved in the dominant phase, like sugar in water. (A popsicle is a good analogy for alloys). When you add sugar to water, you get sugary water. Add more sugar, and you still have water–it’s just sweeter.
But what happens if you add too much sugar? The amount of sugar that can fit into water at 90°C is a lot more than can fit at 10°C. In this case, the excess sugar will precipitate and form sugar crystals–alloy precipitation is exactly the same!
Effects of Solid Solution
Why is alloying two metals so different from combining them in a composite? After all, isn’t it the same amount of atoms?
The answer has to do with bonding. In an alloy the atoms are all mixed together. Every atom from one element interacts with several atoms of the other element. This is a very different situation from when atoms of an element primarily interact with themselves!
If you’re still confused about the difference between an alloy and a metal-metal composite, check out this article.
But why does solid solution mixing actually occur? And why do the atoms arrange randomly, rather than in a specific order?
Advanced explanation of why solid solution mixing occurs
Assume that you mix two elements together: A and B.
It’s possible that A atoms like each other much better than B atoms. In that case, the material will split into two phases (like oil and water). This minimizes the amount of A atoms that touch B atoms.
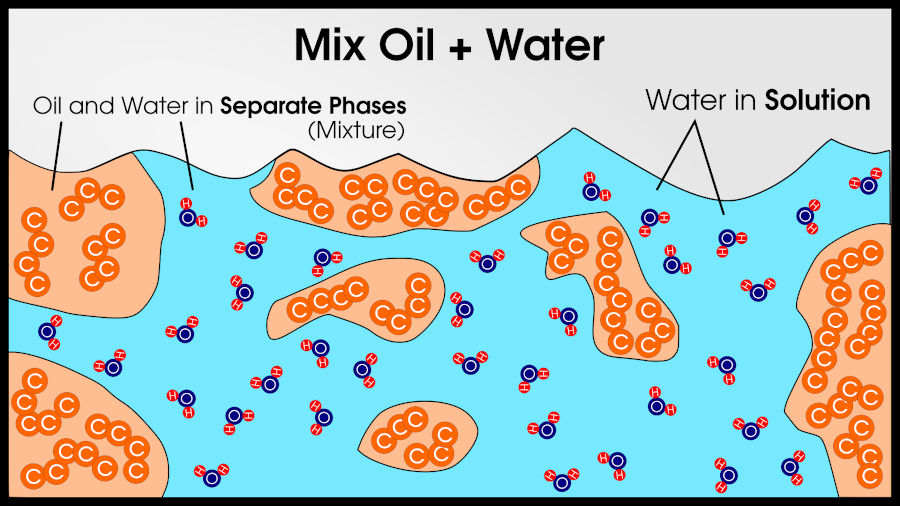
It’s also possible that A atoms like B atoms much more than they like other A atoms. This situation results in an ordered compound (like sodium and chloride). This maximizes the number of A atoms that touch B atoms.
It’s also possible (and quite likely, in metals), that A and B atoms like each about as much as they like themselves. This results in a solid solution (like most metals). If A and B are the same size, they can randomly substitute for each other. If A and B are different sizes, the smaller one rests in interstitial sites between the larger atoms.
The atoms arrange randomly because it maximizes entropy. Entropy is basically chaos, and the universe always tries to increase entropy. That’s the 2nd law of thermodynamics.
This “entropic driving force” can actually change what kind of alloy results. For example, suppose A and B don’t really like each other. Normally, they’d form separate phases. But if there is 99% A and 1% B, there is a huge entropy gain from spreading those B atoms among all the A atoms. So you would probably get a solid solution.
However once there are roughly even amounts of A and B, the entropy gain from spreading B atoms is not large enough to overcome B’s dislike of A. Separate phases result!
This may be a bit hard to understand, so imagine that you have a clean room. You come home, take off your clothes, and throw them on the floor. Your room is much messier than before! But imagine your room is already messy. Does one more dirty shirt really affect the overall messiness? Odds are that by that point, you’ve developed a pile of clothes on the ground and the shirt will go onto the pile.
This is why A and B atoms can enter solid solution even if A-A and B-B bonds are stronger than A-B bonds. For most alloys, in fact, A-A bonds and B-B bonds will be stronger than A-B bonds.
That is why the melting point decreases when you alloy metals. Melting an alloy means adding kinetic energy until the atoms break their bonds. Weaker bonds mean a lower melting point.
But why are alloys stronger than the pure elements?
It’s very difficult to explain this unless you know other basic materials science concepts. For those of you who like a challenge, a brief explanation is included in the collapsable text.
Why alloys are stronger than pure metals
A material’s strength is its resistance to deformation.
Metals move by dislocations, so a stronger metal means it’s more difficult to slide atoms past each other. Although A-B bonds are weaker than A-A or B-B bonds (making sliding easier), the fact that A and B atoms are different sizes means there is a strain field.
This strain field impedes dislocation motion, resulting in an overall stronger metal. We call this solid-solution strengthening, and it is one of the most reliable ways to make stronger metals.
Types of alloys
There are several broad classes of alloys.
Regular alloys
Substitutional or interstitial alloys. Most of the composition is made of a single element, with small additions of other elements. If there are two elements, it’s a “binary alloy,” three elements is “ternary,” four elements is “quaternary,” and five elements is “quinary.” In practice, alloys with more than three different elements will be called “multi-component” alloys.
Steels
Steels are not fundamentally different from other alloys, except that they are perhaps the most advanced material metallurgy can offer. Highly engineered steels may have a dozen elements in small amounts (in addition to Fe and C).

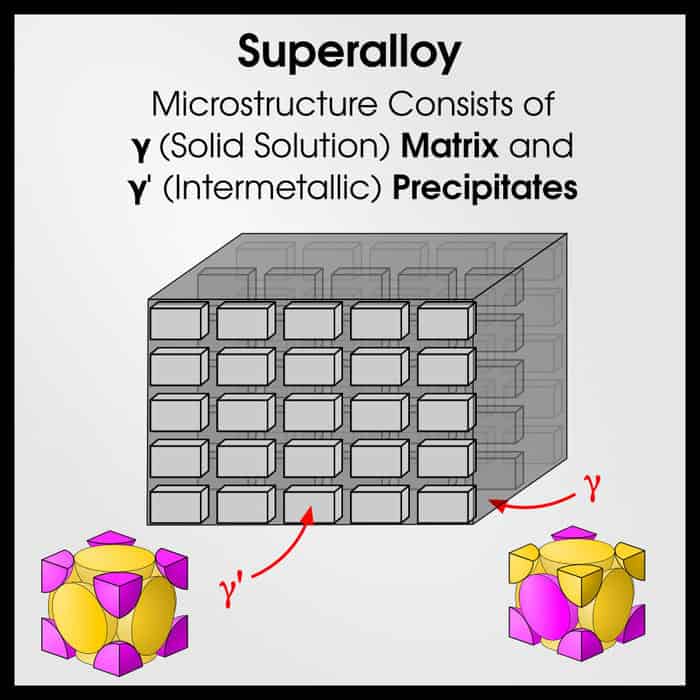
Superalloys
Superalloys are another pinnacle of metallurgy. Superalloys are like micro-scale composites. They have a matrix (solid solution) phase, and a precipitate (intermetallic) phase. Like steels, modern superalloys may have 10-20 alloying elements.
HEAs
High-Entropy Alloys (HEAs) take solid solution strengthening to the extreme. Remember how I said that most alloys are primarily composed of a single element? That’s because intermetallics may form if two elements occur in similar amounts. Instead, HEAs combine 5 or 6 metal elements in equal ratios. Because of the entropy of mixing, the alloy stays as a single phase. HEAs are a new type of alloy and are not commercially widespread.
BMGs
Bulk Metallic Glasses (BMGs) are metals arranged in an amorphous structure. That means there is no orderly crystal structure. These are slightly more developed than HEAs, but they are still rare.
Intermetallics
Intermetallics are not considered alloys by every materials scientist (see my argument here). I prefer to call them “intermetallic compounds” instead of “intermetallic alloys,” because the atoms are arranged in an ordered structure like ceramics, rather than in solid solution. In general, intermetallics have terrible properties and are completely useless. However, there are two notable exceptions. The strengthening phase of superalloys is an intermetallic, and NiTi is an intermetallic shape memory alloy.
Can a Pure Metal be an Alloy?
This is a purely semantic argument. Most materials scientists disagree with me, so reading this will likely confuse you. Still, I want to get it off of my chest so I decided to put it here in this collapsible text.
Click here to expand!
To me, the word “alloy” suggests a compositionally engineered metal. Add carbon to iron, and get the stronger alloy, steel. Add chromium to steel, and get the corrosion-resistant stainless steel.
Alloying a metal means adjusting its composition to achieve certain properties. But can “dealloying” achieve the same thing? I believe it can.
Imagine that I wanted a material with the highest melting point possible. We’ve already discussed that having extra atoms in your metal decreases the melting point. So in this case, I would want to take pure tungsten and purify it further.
“Pure tungsten” may be 99.99% tungsten, and 0.01% other things that the manufacturer couldn’t remove. So what if I intentionally design a process to make my tungsten 99.99999% pure, to achieve the highest melting point possible?
I no longer have a default pure element, I have an intentionally dealloyed material. Does this mean it’s an “alloy” again?
I think so, but so far no one else agrees with me!
Alloy Examples
Alloys can be defined by their patent, historical use, or manufacturing specifications.
These specifications are often developed because certain impurities need to be removed. For example, many steel alloys have 0.03-0.05% phosphorus. This isn’t because adding phosphorus improves the steel–it’s because that’s the maximum acceptable level of phosphorus and it’s too expensive to remove more than that.
Here are a few examples of common or unique alloys.
Sterling silver
Sterling silver is an alloy used in jewelry. In jewelry, it’s defined as having 92.5 wt% silver, with the rest usually being copper. The copper is added to increase the silver’s strength (pure silver is usually too soft to wear as jewelry). As a trade-off, sterling silver has lower oxidation/corrosion resistance than pure silver.
Ti-6Al-4V
Ti64 (pronounced Tie 6 4) is the most-used titanium alloy. It has excellent strength-to-density, very good corrosion and oxidation resistance, and is mostly biocompatible. Ti64 dominates the aerospace industry and so much is produced that– even though there are more biocompatible alloys–Ti64 is also frequently used in the medical field for implants. Any time you see the buzzword “made of titanium” such as on the SR-71, they probably mean Ti-6Al-4V.

Ni-based superalloys
Superalloys can operate at a high fraction of their melting temperature. They have great resistance to creep, oxidation, and resistance due to the unique microstructure. They are relatively expensive to make, so Ni-based superalloys are mostly used as blades in the hottest sections of a turbine engine.

Stainless steel
Stainless Steel is a steel alloy (iron and carbon) with chromium, which provides oxidation and corrosion resistance. This is useful whenever you don’t want steel to rust such as kitchen utensils, surgical instruments, water pipes, and jewelry.
Solder
Solders are low melting point, conductive alloys. They are usually made of lead, tin, and/or bismuth. Because they have a low melting point, plumbers, jewelers, and electricians melt solders between other metals to join them together (like hot glue). Solders are most well-known for connecting wires to a circuit board.

Bronze
Bronzes are copper alloys. Apart from copper, they consist of tin or other elements like aluminum, silicon, nickel. They are usually used for their combination of strength and high corrosion resistance.

Brass
Brass is another commonly-used copper alloy. They consist of copper and zinc. Brasses are widely used in everyday applications, for example as jewelry, coins, musical instruments, cartridge casings, etc.

Bulk Metallic Glass
Vitreloy was the first (and currently only?) commercial bulk metallic glass (BMG). That means that the atoms are not in a crystal structure, but are disorganized like regular glasses. This increases corrosion resistance, strength, and elasticity, but almost completely eliminates ductility at room temperature. BMGs are often used in sporting equipment because they are very stiff.
NiTi
50% Ni and 50% Ti forms an intermetallic compound that is biocompatible, extremely elastic, and can restore its shape when heated. Normally, intermetallic compounds are very strong and brittle because dislocations are blocked, but NiTi can deform by twinning instead. This is also what allows the shape memory process. NiTi is used in wire glasses frames, braces wires, and stents.
Final Thoughts
Alloys are some of the most important engineering materials. Metals are useful for their conductivity, reflectivity, formability, and warning signs that they will break. They are relatively cheap to mine and manufacture.
Almost all real-world metals are alloys, instead of pure metals. Among other advantages, alloys are usually stronger and have a lower melting point than pure metals. The most important downside of alloys is that they have lower conductivity than pure metals–which is why the only pure metal you encounter in daily life is copper in electrical wires.