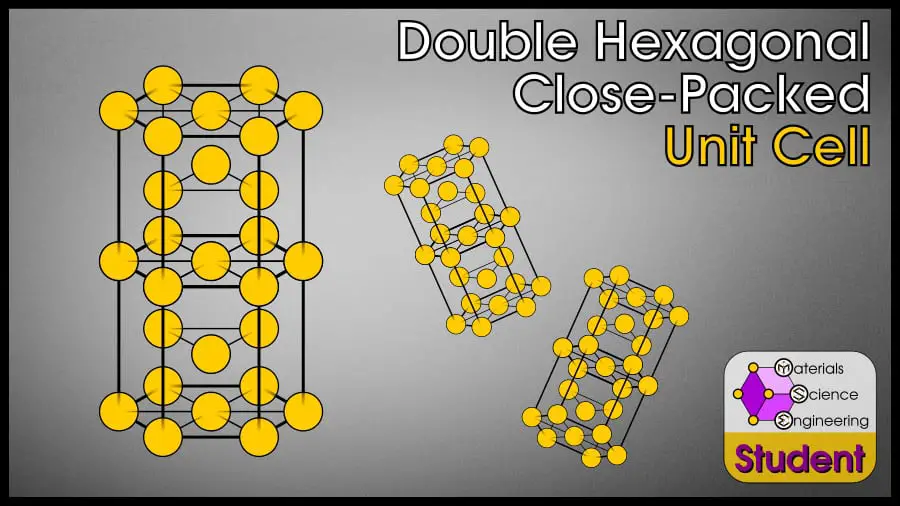
The Double Hexagonal Close-Packed (DHCP) crystal structure is another close-packed, hexagonal crystal structure; however, this is not a very common crystal structure, so I’m assuming if you’re looking for information on DHCP, you’re a somewhat advanced student in materials science. I’ll give you the facts quickly and with little explanation, but if you are new to materials science and don’t know what something means, you can check out my articles on the FCC, BCC, or HCP crystals, which explain terminology more slowly.
The DHCP crystal structure is similar to FCC or HCP, but it uses a 4-layer stacking pattern ABAC-ABAC, instead of FCC’s ABC-ABC or HCP’s AB-AB stacking pattern. The double hexagonal close-packed cell belongs to space group #194 or P63/mmc, Strukturbericht A3’, and Pearson symbol hP4. α-La is the prototype for DHCP.
The Double Hexagonal Close-Packed (DHCP) unit cell can be imagined as two HCP cells stacked on top of each other, but the middle layer alternates between B and C. It may also be known as a La-type crystal, because lanthanum is the first element to display this structure. DHCP has 12 atoms per conventional unit cell, lattice constant and
(or
), coordination number CN=12, and Atomic Packing Factor APF=0.74. DHCP is a close-packed structure with ABAC-ABAC stacking.
Outline
Common Examples of Double Hexagonal Close-Packed Materials
Although there are quite a few elements with DHCP structures, they are mostly heavy elements that are not often used in introductory materials science. In addition to lanthanum (remember, DCHP may sometimes be called the La-type structure), praseodymium, neodymium, promethium, cerium, americium, curium, berkelium, and californium also have the DHCP crystal structure.
Many of these elements are radioactive, which may help explain why they form DHCP instead of HCP or FCC. Nonetheless, the DHCP phase is one of the more-stable crystal structures because of its high coordination number.
Double Hexagonal Close-Packed Coordination Number
Since you know that DHCP is close-packed, I’m sure you figured out that each atom has 12 nearest neighbors (NNs), and therefore CN=12.
Double Hexagonal Close-Packed Lattice Constants
As with HCP, DHCP has two lattice constants: a and c.
Lattice parameters a and b are the same length (twice the atomic radius) and at 120º to each other. Lattice parameter c extends in the vertical direction, and is longer than a.
By convention, it’s common to describe the c/a ratio, rather than c by itself.
If you know how to calculate the c/a ratio for HCP, it should be easy to calculate the c/a ratio for DHCP. In fact, it’s just double, since DHCP is effectively 2 HCP cells stacked on top of each other. Because of this, you may also see the DHCP lattice parameters expressed as a and .
Therefore, the lattice constant and
. Or,
, or
.
These are theoretical values, however, calculated from the hard sphere model. Structures usually take the DHCP (or even HCP) structures because they have some slight asymmetry in the c-direction, so the c/a ratio is usually not ideal.
If you wanted to describe the DHCP crystal with math, you would describe the cell with the primitive lattice vectors:
The basis vectors are:
Double Hexagonal Close-Packed Atomic Packing Factor
I’m sure it will not surprise you that the DHCP APF is 74%, the same as any other close-packed structure. Of course, that’s the ideal APF, but as I just mentioned, DHCP crystals rarely have ideal c/a ratios.
If you know the specific a parameter and c/a ratio of your DHCP crystal, here is how to calculate the Atomic Packing Factor (APF).
First, you need to calculate the volume of the unit cell. Let’s start by breaking it into a hexagon and a height.
The hexagon is 6 equilateral triangles, with side length a. Using the proper formula (or clever trigonometry), you’d find that the area of an equilateral triangle is . Six of those gives us our hexagon, and the height is c.
Working this out, we have , for the volume of the hexagonal prism in terms of a and c/a.
Now we need to determine the volume taken up by atoms.
DHCP has 12 vertex atoms, which each contribute ⅙ of their total volume, for a total of 2 whole atoms. There are 2 face atoms which each contribute ½ of their total volume, for one whole atom. There are 6 edge atoms which each contribute ⅓ of their total volume for 2 whole atoms. Finally, there are 7 atoms completely inside the cell, which contribute their full volume.
If you add this up, you find that there are 12 atoms inside this volume. With the hard sphere model, each atom has a volume of .
Thus, the APF for DHCP is
Where is the radius of the atom,
is the lattice parameter
, and
is the c/a ratio.
And of course if you simplify this with and
, you end up with the expected APF=74%.
Final Thoughts
The Double Hexagonal Close-Packed crystal structure is a surprisingly common crystal structure that is rarely talked about in materials science classes. This close-packed structure appears in several lanthanides and actides. DHCP has ABAC-ABAC stacking, has opposed to more common AB-AB (HCP) and ABC-ABC (FCC) stacking.
It is worth noting that while this page listed several theoretical values for abstract DHCP crystals, real DHCP crystals vary in their c/a ratios.
References and Further Reading
If you want to know more about the basics of crystallography, check out this article.
If you weren’t sure about the difference between crystal structure and Bravais lattice, check out this article.
I also mentioned atomic packing factor (APF) earlier in this article. This is an important concept in your introductory materials science class, so if you want a full explanation of APF, check out this page.
If you’re interested in advanced crystallography or crystallography databases, you may want to check out the AFLOW crystallographic library.
For a great reference for all crystal structures, check out “The AFLOW Library of Crystallographic Prototypes.”
Single-Element Crystal Structures and the 14 Bravais Lattices
If you want to learn about specific crystal structures, here is a list of my articles about Bravais lattices and some related crystal structures for pure elements.
1. Simple Cubic
2. Face-Centered Cubic
2a. Diamond Cubic
3. Body-Centered Cubic
4. Simple Hexagonal
4a. Hexagonal Close-Packed
4b. Double Hexagonal Close-Packed (La-type)
5. Rhombohedral
5a. Rhombohedral Close-Packed (Sm-type)
6. Simple Tetragonal
7. Body-Centered Tetragonal
7a. Diamond Tetragonal (White Tin)
8. Simple Orthorhombic
9. Base-Centered Orthorhombic
10. Face-Centered Orthorhombic
11. Body-Centered Orthorhombic
12. Simple Monoclinic
13. Base-Centered Monoclinic
14. Triclinic
Other articles in my crystallography series include:
Introduction to Bravais Lattices
What is the Difference Between “Crystal Structure” and “Bravais Lattice”
Atomic Packing Factor
How to Read Miller Indices
How to Read Hexagonal Miller-Bravais Indices
Close-Packed Crystals and Stacking Order
Interstitial Sites
Primitive Cells
How to Read Crystallography Notation
What are Point Groups
List of Point Groups
What are Space Groups
List of Space Groups
The 7 Crystal Systems